Lewis Acid
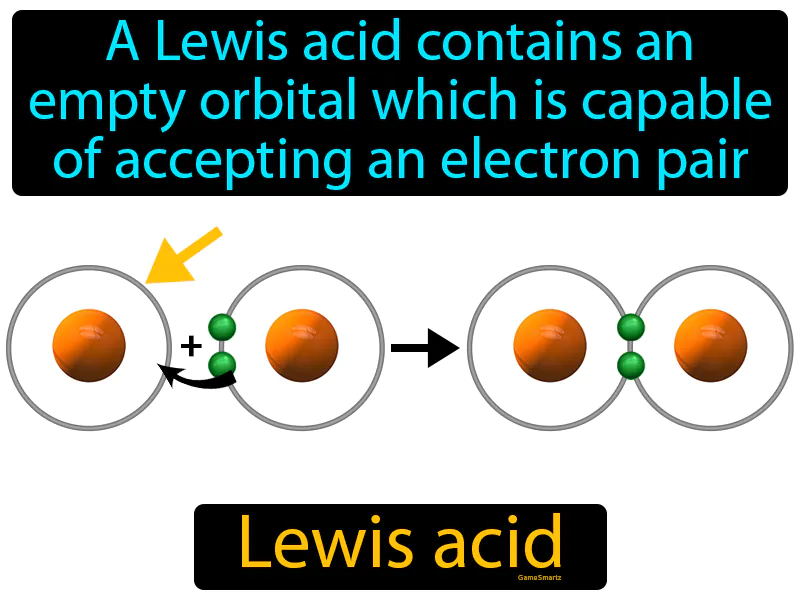
Imagine trying to find a parking spot in a crowded lot with one empty space left. Just like that empty parking space is waiting for a car to fill it, a Lewis acid has an empty orbital ready to accept an electron pair. In this analogy, the parking space represents the empty orbital, and the car represents the electron pair; both are seeking a spot where they can fit perfectly, completing a necessary connection.
Practice Version
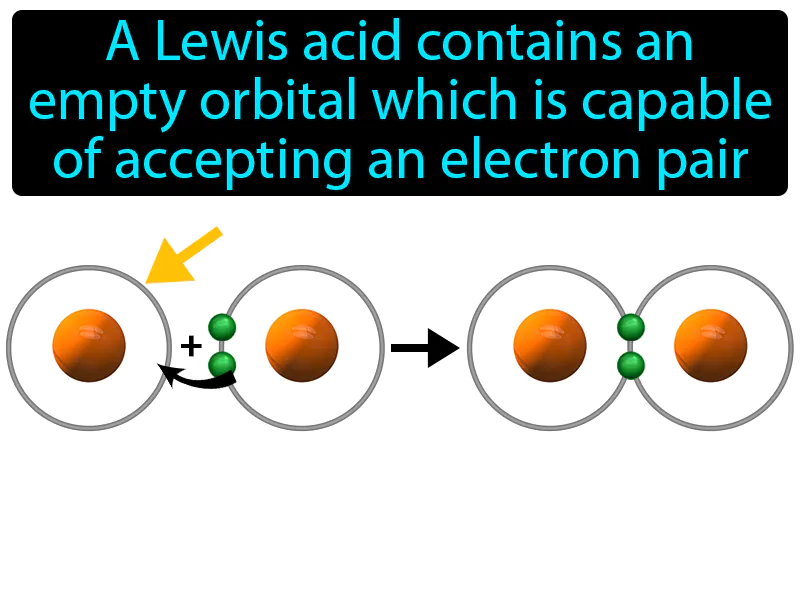
Lewis Acid: A Lewis acid contains an empty orbital which is capable of accepting an electron pair. Lewis acid. In simple terms, a Lewis acid is a substance that can accept a pair of electrons from another molecule.