Hydronium Ion
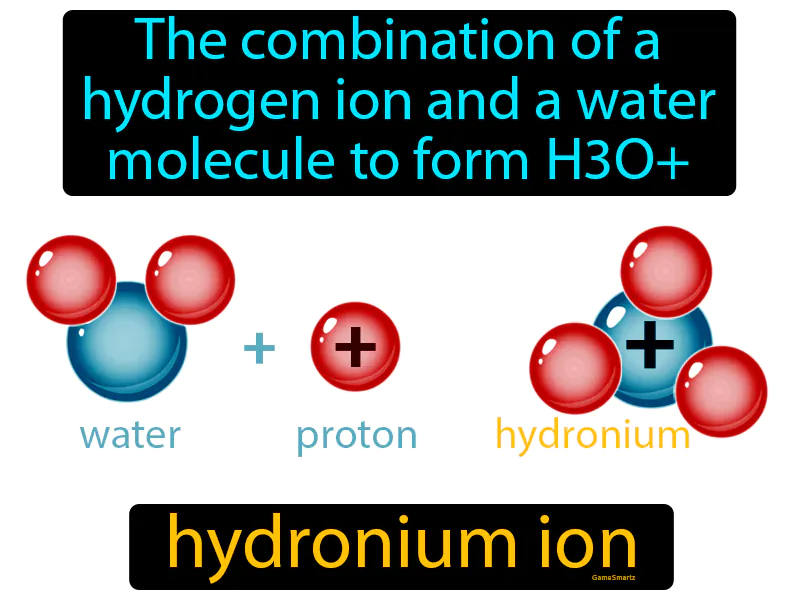
Imagine you're at a crowded party, trying to find a friend, but you end up bumping into a stranger and striking up a conversation. Just like how you and the stranger connect and form a new social interaction, a hydrogen ion bumps into a water molecule, forming a hydronium ion (H3O+). In this analogy, you are the hydrogen ion, the stranger represents the water molecule, and your new conversation is akin to the formation of the hydronium ion, as both involve two separate entities coming together to create something new.
Practice Version
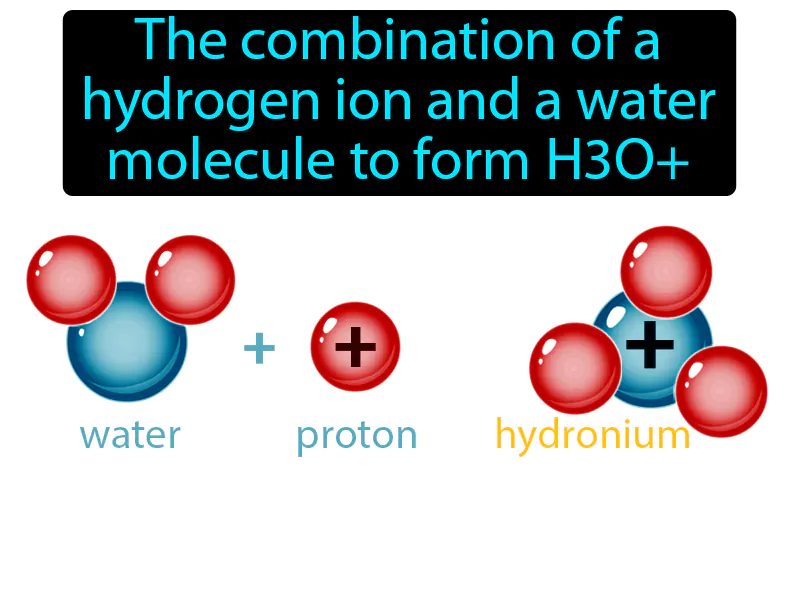
Hydronium Ion: The combination of a hydrogen ion and a water molecule to form H3O hydronium ion. The hydronium ion is a water molecule with an added hydrogen ion, making it an important player in determining the acidity of solutions.